Abstract
Keywords
1. Introduction
2. Methods
2.1. Epidemiological model
Table 1. Epidemiological model parameters for SIV-A.
| Symbol | Description | Baseline value | Range* | Source |
|---|---|---|---|---|
| N | Average number of pigs in a wean-to-finish commercial farm | 2500 | (Cornelison et al., 2018) | |
| R0 | Basic reproduction number | 6 | 2.5–10.66 | (Allerson et al., 2013, Romagosa et al., 2011, Rose et al., 2013) |
| β | Transmission rate (per day) | (Keeling and Rohani, 2007, M May, 1991) | ||
| V | Vaccine efficacy | varied | 60 % – 100 % | (Allerson et al., 2013, Romagosa et al., 2011, Vincent et al., 2010) |
| 1/σ | Latency period (days) | 2 | 1.4–5.0 | (Romagosa et al., 2012, Rose et al., 2013) |
| 1/γ | Infection duration (days) | 5 | 2.4–10.4 | (Romagosa et al., 2012, Rose et al., 2013) |
- *
-
For the uncertainty and sensitivity analysis, we used uniform distributions across parameter range values to account for the impact of parameter variability on the robustness of our model outcomes. The uncertainty analysis provides information on the mean and 95 % confidence interval values of the model outcome.
2.2. Economic Model
2.2.1. Benefit-cost analysis
Table 2. Costs associated with influenza infection in swine from the literature.
| Parameter | Average | Range | Source |
|---|---|---|---|
| Feed penalty | 4.93 kg | 2.3–9.8 kg | (Er et al., 2016, Er et al., 2014, Er, 2024) |
| Feed cost (US$) per kilogram | $0.29 | $0.19 – $0.39 | (Cornelison et al., 2018) |
| Per head cost of raising pigs (US$) | $174.36 | $152.24 – $203.50 | (Estimated Livestock Returns [WWW Document], 2025) |
| Influenza-induced mortality | 2.2 % | 1.5–3.30 % | (Papatsiros et al., 2023, Schaefer et al., 2015) |
| Per-head lost profit (US$) | $12.86 | $3.35-$29.26 | (Estimated Livestock Returns [WWW Document], 2025) |
| Per-head carcass disposal (US$) | $9.07 | $7.34–10.66 | (Estimated Livestock Returns [WWW Document], 2025) |
2.2.2. Cost information
2.2.3. Uncertainty analysis
2.2.4. Sensitivity analysis
2.2.5. Simulation
3. Results
3.1. Epidemiological model
Table 3. Average attack rate and 95 % confidence interval of the stochastic simulations of the epidemiological model.
| Vaccines Efficacy | Mean | 95 % Confidence Interval |
|---|---|---|
| 60 % | 50.69 % | (95 % CI:48.98 %, 52.40 %) |
| 50 % | 31.27 % | (95 % CI: 29.85 %, 32.69 %) |
| 70 % | 4.71 % | (95 % CI: 4.29 %, 5.14 %) |
| 80 % | 0.10 % | (95 % CI: 0.10 %, 0.11 %) |
| 90 % | 0.06 % | (95 % CI: 0.053, 0.057) |

Fig. 1. Median and 95 % confidence interval of the stochastic simulations of the epidemiological model by vaccine efficacy.
3.2. Economic evaluation
Table 4. Net Present Value per pig in USD$ by vaccine efficacy and total cost of vaccination per pig. Values are presented as mean and 95 % Confidence Interval.
| Empty Cell | Total vaccination cost per pig | ||||
|---|---|---|---|---|---|
| Efficacy | $1.34 | $1.68 | $2.02 | $2.36 | $2.70 |
| 60 % | -1.26 (-1.48, −1.04) |
-1.60 (-1.82, −1.38) |
-1.94 (-2.16, −1.72) |
-2.28 (-2.50, −2.06) |
-2.62 (-2.84, −2.40) |
| 70 % | -0.41 (-0.57, −0.25) |
-0.75 (-0.91, –0.59) |
-1.09 (-1.25, −0.93) |
-1.43 (-1.59, −1.27) |
-1.77 (-1.93, −1.61) |
| 80 % | 2.26 (2.15, 2.38) |
1.92 (1.81, 2.04) |
1.58 (1.47, 1.70) |
1.24 (1.13, 1.36) |
0.90 (0.79, 1.02) |
| 90 % | 3.39 (3.38, 3.40) |
3.05 (3.04, 3.06) |
2.71 (2.70, 2.72) |
2.37 (2.36, 2.38) |
2.03 (2.02, 2.04) |
| 95 % | 3.41 (3.40, 3.41) |
3.07 (3.06, 3.07) |
2.73 (2.72, 2.73) |
2.39 (2.38, 2.39) |
2.05 (2.04, 2.05) |
Table 5. Benefit Cost Ratio by vaccine efficacy and total cost of vaccination per pig. Values are presented as mean and 95 % Confidence Interval for the Monte Carlo Simulation.
| Empty Cell | Total vaccination cost per pig | ||||
|---|---|---|---|---|---|
| Efficacy | $1.34 | $1.68 | $2.02 | $2.36 | $2.70 |
| 60 % | 0.069 (0.067, 0.071) |
0.055 (0.053, 0.056) |
0.046 (0.044, 0.047) |
0.039 (0.038, 0.040) |
0.034 (0.033, 0.035) |
| 70 % | 0.800 (0.799, 0.082) |
0.638 (0.637,0.639) |
0.531 (0.530, 0.532) |
0.454 (0.453, 0.455) |
0.397 (0.396, 0.398) |
| 80 % | 3.102 (3.101, 3.103) |
2.474 (2.473, 2.475) |
2.057 (2.056, 2.058) |
1.761 (1.761, 1.762) |
1.539 (1.538, 1.539) |
| 90 % | 4.068 (4.065, 4.071) |
3.247 (3.245, 3.250) |
2.701 (2.699, 2.703) |
2.310 (2.308, 2.311) |
2.020 (2.018, 2.021) |
| 95 % | 4.087 (4.082, 4.092) |
3.257 (3.252, 3.261) |
2.711 (2.708, 2.715) |
2.319 (2.316, 2.322) |
2.028 (2.026, 2.031) |

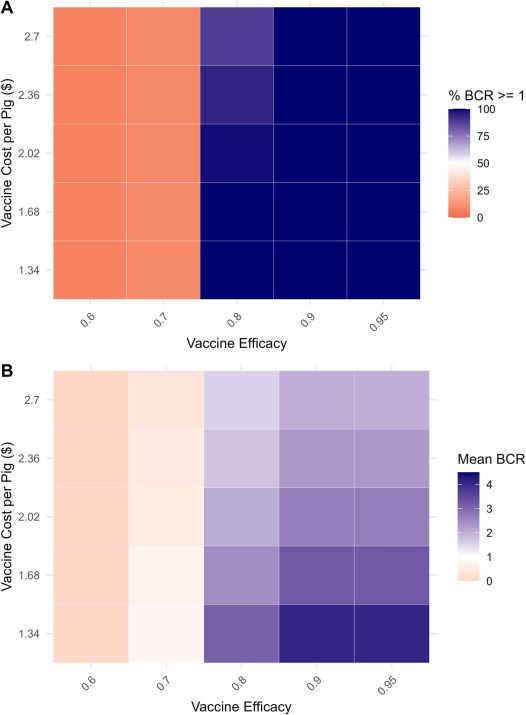
Fig. 2. A) Percent of Monte Carlo simulations with a BCR greater than 1 by vaccine efficacy and cost of implementing the strategy per pig. B) Average BCR of Monte Carlo simulations by vaccine efficacy and cost of implementing the strategy per pig.
3.3. Sensitivity analysis

Fig. 3. Tornado plot of standardized regression coefficients from a linear model predicting the benefit-cost ratio (BCR) of influenza vaccination in swine relative to the baseline scenario. Each bar represents the relative importance of one input parameter on BCR, controlling for the others.
4. Discussion
Funding
CRediT authorship contribution statement
Declaration of Competing Interest
Data availability
References
- Allerson et al., 2013
The impact of maternally derived immunity on influenza A virus transmission in neonatal pig populationsVaccine, 31 (2013), pp. 500-505, 10.1016/j.vaccine.2012.11.023
- Almeida et al., 2017
A cross-sectional study of swine influenza in intensive and extensive farms in the northeastern region of the state of São Paulo, BrazilTrop. Anim. Health Prod., 49 (2017), pp. 25-30, 10.1007/s11250-016-1153-z
- Alvarez et al., 2015
Association of the presence of influenza A virus and porcine reproductive and respiratory syndrome virus in sow farms with post-weaning mortalityPrev. Vet. Med., 121 (2015), pp. 240-245, 10.1016/j.prevetmed.2015.07.003
- Andraud et al., 2023
Evaluation of early single dose vaccination on swine influenza A virus transmission in piglets: From experimental data to mechanistic modellingVaccine, 41 (2023), pp. 3119-3127, 10.1016/j.vaccine.2023.04.018
- Augusto et al., 2020
Effect of porcine reproductive and respiratory syndrome (PRRS) modified-live virus (MLV) interventions on growth performance of infected growing pigs under field conditionsIowa State University (2020)
- Avanthay et al., 2024
Evaluation of a novel intramuscular prime/intranasal boost vaccination strategy against influenza in the pig modelPLoS Pathog., 20 (2024), Article e1012393, 10.1371/journal.ppat.1012393
- Boardman et al., 2018
Cost-benefit analysis (5th ed), Cambridge University Press, Cambridge, England (2018), 10.1017/9781108235594
- Briggs et al., 2012
Model parameter estimation and uncertainty: a report of the ISPOR-SMDM Modeling Good Research Practices Task Force–6Value Health, 15 (2012), pp. 835-842, 10.1016/j.jval.2012.04.014
- Brookes et al., 2010
Replication, pathogenesis and transmission of pandemic (H1N1) 2009 virus in non-immune pigsPLoS One, 5 (2010), Article e9068, 10.1371/journal.pone.0009068
- Brown, 2000
The epidemiology and evolution of influenza viruses in pigsVet. Microbiol, 74 (2000), pp. 29-46, 10.1016/s0378-1135(00)00164-4
- Brown, 2013
History and Epidemiology of Swine Influenza in EuropeJ.A. Richt, R.J. Webby (Eds.), Swine Influenza, Springer Berlin Heidelberg, Berlin, Heidelberg (2013), pp. 133-146, 10.1007/82_2011_194
- Cador et al., 2016a
Maternally-derived antibodies do not prevent transmission of swine influenza A virus between pigsVet. Res., 47 (2016), p. 86, 10.1186/s13567-016-0365-6
- Cador et al., 2016b
Maternally Derived Immunity Extends Swine Influenza A Virus Persistence within Farrow-to-Finish Pig Farms: Insights from a Stochastic Event-Driven Metapopulation ModelPLoS One, 11 (2016), Article e0163672, 10.1371/journal.pone.0163672
- Cador et al., 2017
Control of endemic swine flu persistence in farrow-to-finish pig farms: a stochastic metapopulation modeling assessmentVet. Res., 48 (2017), p. 58, 10.1186/s13567-017-0462-1
- Calderón Díaz et al., 2020
Financial analysis of herd status and vaccination practices for porcine reproductive and respiratory syndrome virus, swine influenza virus, and Mycoplasma hyopneumoniae in farrow-to-finish pig farms using a bio-economic simulation modelFront. Vet. Sci., 7 (2020), Article 556674, 10.3389/fvets.2020.556674
- Carnell, 2022
tornado: Plots for Model Sensitivity and Variable ImportanceCRAN Contrib. Packages (2022), 10.32614/cran.package.tornado
- Carnell, 2025
Carnell, R., 2025. Plots for Model Sensitivity and Variable Importance [WWW Document]. URL 〈https://cran.r-project.org/web/packages/tornado/tornado.pdf〉 (accessed 1.27.26).
- CDC, 2024
CDC, 2024. About Influenza in Swine (Pigs) [WWW Document]. Swine / Variant Influenza. URL 〈https://www.cdc.gov/swine-flu/about/influenza-in-swine.html〉 (accessed 1.26.26).
- Cornelison et al., 2018
Impact of health challenges on pig growth performance, carcass characteristics, and net returns under commercial conditionsTransl. Anim. Sci., 2 (2018), pp. 50-61, 10.1093/tas/txx005
- Corzo et al., 2013
Airborne detection and quantification of swine influenza a virus in air samples collected inside, outside and downwind from swine barnsPLoS One, 8 (2013), Article e71444, 10.1371/journal.pone.0071444
- CPI Inflation Calculator [WWW Document], 2025
CPI Inflation Calculator [WWW Document], 2025. Bureau of Labor Statistics. URL 〈https://www.bls.gov/data/inflation_calculator.htm〉 (accessed 6.16.25).
- DeGraves and Fetrow, 1991
Partial budget analysis of vaccinating dairy cattle against coliform mastitis with an Escherichia coli J5 vaccineJ. Am. Vet. Med. Assoc., 199 (1991), pp. 451-455
- Detmer et al., 2013
In vivo evaluation of vaccine efficacy against challenge with a contemporary field isolate from the α cluster of H1N1 swine influenza virusCan. J. Vet. Res., 77 (2013), pp. 24-32
- Er et al., 2014
Adverse effects of Influenza A(H1N1)pdm09 virus infection on growth performance of Norwegian pigs – a longitudinal study at a boar testing stationBMC Vet. Res., 10 (2014), p. 284, 10.1186/s12917-014-0284-6
- Er et al., 2016
Production impact of influenza A(H1N1)pdm09 virus infection on fattening pigs in NorwayJ. Anim. Sci., 94 (2016), pp. 751-759, 10.2527/jas.2015-9251
- Er, 2021
Longitudinal Projection of Herd Prevalence of Influenza A(H1N1)pdm09 Virus Infection in the Norwegian Pig Population by Discrete-Time Markov Chain ModellingInfect. Dis. Rep., 13 (2021), pp. 748-756, 10.3390/idr13030070
- Er, 2024
A Norwegian observational study of feed conversion efficiency in Duroc and Landrace grower pigs seropositive for influenza A (H1N1) pdm09 virusJ. Swine Health Prod. (2024), 10.54846/jshap/1395
- Estimated Livestock Returns [WWW Document], 2025
Estimated Livestock Returns [WWW Document], 2025. Estimated Livestock Returns. URL 〈https://estimatedreturns.econ.iastate.edu/〉 (accessed 6.13.25).
- Etbaigha et al., 2018
An SEIR model of influenza A virus infection and reinfection within a farrow-to-finish swine farmPLoS One, 13 (2018), Article e0202493, 10.1371/journal.pone.0202493
- Fast et al., 2015
Cost-effective control of infectious disease outbreaks accounting for societal reactionPLoS One, 10 (2015), Article e0136059, 10.1371/journal.pone.0136059
- Fiatsonu et al., 2024
Assessing the impact of a potential canine vaccine for the control of Chagas disease: a mathematical modeling studyFront. Trop. Dis., 5 (2024), 10.3389/fitd.2024.1421669
- FluSure XP Product Sheet [WWW Document], n.d
FluSure XP Product Sheet [WWW Document], n.d. URL 〈https://www.zoetisus.com/content/_assets/docs/vmips/package-inserts/flusure-xp-product-information.pdf〉 (accessed 11.28.16).
- Flusure XP Swine Vaccine [WWW Document], 2025
Flusure XP Swine Vaccine [WWW Document], 2025. URL 〈https://pipevet.com/flusure-xp-swine-vaccine〉 (accessed 12.23.25).
- Gauger et al., 2012
Kinetics of lung lesion development and pro-inflammatory cytokine response in pigs with vaccine-associated enhanced respiratory disease induced by challenge with pandemic (2009) A/H1N1 influenza virusVet. Pathol., 49 (2012), pp. 900-912, 10.1177/0300985812439724
- Gebhardt et al., 2020
Postweaning mortality in commercial swine production. I: review of non-infectious contributing factorsTransl. Anim. Sci., 4 (2020), Article txaa068, 10.1093/tas/txaa068
- Gumbert et al., 2020
Reproductive performance of pandemic influenza A virus infected sow herds before and after implementation of a vaccine against the influenza A (H1N1)pdm09 virusPorc. Health Manag, 6 (2020), p. 4, 10.1186/s40813-019-0141-x
- Haden et al., 2012
Assessing production parameters and economic impact of swine influenza, PRRS and Mycoplasma hyopneumoniae on finishing pigs in a large production systemI.A. Perry (Ed.), Presented at the American Association of Swine Veterinarians Annual Meeting (2012), pp. 75-76
- Haley, 2025
Haley, M., 2025. Hogs & Pork – Sector at a Glance [WWW Document]. URL 〈https://www.ers.usda.gov/topics/animal-products/hogs-pork/sector-at-a-glance〉 (accessed 1.27.26).
- Horst et al., 1999
Monte Carlo simulation of virus introduction into the NetherlandsPrev. Vet. Med., 41 (1999), pp. 209-229, 10.1016/s0167-5877(99)00038-0
- Janzen et al., 2025
Sources and sinks of influenza A virus genomic diversity in swine from 2009 to 2022 in the United StatesJ. Virol., 99 (2025), Article e0054125, 10.1128/jvi.00541-25
- Johnson, 2010
adaptivetau: Tau-Leaping Stochastic SimulationCRAN Contrib. Packages (2010), 10.32614/cran.package.adaptivetau
- Karsten et al., 2005
Monte Carlo simulation of classical swine fever epidemics and controlI. Gen. Concepts Descr. Model. Vet. Microbiol, 108 (2005), pp. 187-198, 10.1016/j.vetmic.2005.04.009
- Keeling and Rohani, 2007
Modeling infectious diseases in humans and animalsPrinceton University Press, Princeton, NJ (2007)
- Kontowicz et al., 2023
A stochastic compartmental model to simulate intra- and inter-species influenza transmission in an indoor swine farmPLoS One, 18 (2023), Article e0278495, 10.1371/journal.pone.0278495
- Le Sage et al., 2024
Potential pandemic risk of circulating swine H1N2 influenza virusesNat. Commun., 15 (2024), p. 5025, 10.1038/s41467-024-49117-z
- Lothan et al., 2022
Country versus pharmaceutical company interests for hepatitis C treatmentHealth Care Manag. Sci., 25 (2022), pp. 725-749, 10.1007/s10729-022-09607-2
- Lyoo et al., 2014
Comparative pathology of pigs infected with Korean H1N1, H1N2, or H3N2 swine influenza A virusesVirol. J., 11 (2014), p. 170, 10.1186/1743-422X-11-170
- M May, 1991
Infectious diseases of humans: dynamics and controlOxford University Press, Oxford (1991)
- Ma and Earn, 2006
Generality of the final size formula for an epidemic of a newly invading infectious diseaseBull. Math. Biol., 68 (2006), pp. 679-702, 10.1007/s11538-005-9047-7
- Ma, 2020
Swine influenza virus: Current status and challengeVirus Res, 288 (2020), Article 198118, 10.1016/j.virusres.2020.198118
- Mancera Gracia et al., 2020
Influenza A Virus in Swine: Epidemiology, Challenges and Vaccination StrategiesFront Vet. Sci., 7 (2020), p. 647, 10.3389/fvets.2020.00647
- Mayo, 2024
Mayo, D., 2024. 2024 Average Farmland Rental Rates and Worker Wages Summary [WWW Document]. URL 〈https://nwdistrict.ifas.ufl.edu/phag/2024/12/20/2024-average-farmland-rental-rates-and-worker-wages-summary/〉 (accessed 12.26.25).
- McInerney et al., 1992
A framework for the economic analysis of disease in farm livestockPrev. Vet. Med., 13 (1992), pp. 137-154, 10.1016/0167-5877(92)90098-z
- McKinnell et al., 2015
Cost-benefit analysis from the hospital perspective of universal active screening followed by contact precautions for methicillin-resistant Staphylococcus aureus carriersInfect. Control Hosp. Epidemiol., 36 (2015), pp. 2-13, 10.1017/ice.2014.1
- MetaFarms and National Pork Board, 2023
MetaFarms & National Pork Board, 2023. Production AnalysisSummary for U.S. PorkIndustry: 2019-2023 [WWW Document]. URL 〈https://porkcheckoff.org/wp-content/uploads/2024/07/2024-Production-Analysis-Full-Report-1.pdf〉 (accessed 12.26.25).
- Moraes et al., 2023
Veterinarian perceptions and practices in prevention and control of influenza virus in the Midwest United States swine farmsFront. Vet. Sci., 10 (2023), Article 1089132, 10.3389/fvets.2023.1089132
- Moura et al., 2022
Comparison of virus detection, productivity, and economic performance between lots of growing pigs vaccinated with two doses or one dose of PRRS MLV vaccine, under field conditionsPrev. Vet. Med., 204 (2022), Article 105669, 10.1016/j.prevetmed.2022.105669
- Nathues et al., 2018
Modelling the economic efficiency of using different strategies to control Porcine Reproductive & Respiratory Syndrome at herd levelPrev. Vet. Med., 152 (2018), pp. 89-102, 10.1016/j.prevetmed.2018.02.005
- Ouyang et al., 2019
Co-infection of swine with porcine Circovirus type 2 and other swine virusesViruses, 11 (2019), p. 185, 10.3390/v11020185
- Papatsiros et al., 2023
Seroprevalence of swine influenza A virus (swIAV) infections in commercial farrow-to-finish pig farms in GreeceVet. Sci., 10 (2023), p. 599, 10.3390/vetsci10100599
- Parys et al., 2022
Alternating 3 different influenza vaccines for swine in Europe for a broader antibody response and protectionVet. Res., 53 (2022), p. 44, 10.1186/s13567-022-01060-x
- Pedersen, 2019
patchwork: The Composer of PlotsCRAN Contrib. Packages (2019), 10.32614/cran.package.patchwork
- Petro-Turnquist et al., 2024
Swine influenza A virus: challenges and novel vaccine strategiesFront. Cell. Infect. Microbiol, 14 (2024), Article 1336013, 10.3389/fcimb.2024.1336013
- Pittman Ratterree et al., 2024
Mechanistic models of influenza transmission in commercial swine populations: A systematic reviewPathogens, 13 (2024), p. 746, 10.3390/pathogens13090746
- Pitzer et al., 2016
High turnover drives prolonged persistence of influenza in managed pig herdsJ. R. Soc. Interface, 13 (2016), 10.1098/rsif.2016.0138
- Plastina, 2024
Plastina, A., 2024. Financial Performance Measures for Iowa Farms [WWW Document]. URL 〈https://www.extension.iastate.edu/agdm/wholefarm/html/c3-55.html〉 (accessed 1.7.26).
- Rajao et al., 2023
Live attenuated influenza A virus vaccine expressing an IgA-inducing protein protects pigs against replication and transmissionFront. Virol., 3 (2023), 10.3389/fviro.2023.1042724
- Report-Texas Daily Grain Bids [WWW Document], 2025
Report-Texas Daily Grain Bids [WWW Document], 2025. URL 〈https://mymarketnews.ams.usda.gov/viewReport/2711〉 (accessed 6.23.25).
- Reynolds et al., 2014
Mathematical modeling of influenza A virus dynamics within swine farms and the effects of vaccinationPLoS One, 9 (2014), Article e106177, 10.1371/journal.pone.0106177
- Romagosa et al., 2011
Vaccination of influenza a virus decreases transmission rates in pigsVet. Res., 42 (2011), p. 120, 10.1186/1297-9716-42-120
- Romagosa et al., 2012
Sensitivity of oral fluids for detecting influenza A virus in populations of vaccinated and non-vaccinated pigsInfluenza Other Respi. Virus, 6 (2012), pp. 110-118, 10.1111/j.1750-2659.2011.00276.x
- Rose et al., 2013
Dynamics of influenza A virus infections in permanently infected pig farms: evidence of recurrent infections, circulation of several swine influenza viruses and reassortment eventsVet. Res., 44 (2013), p. 72, 10.1186/1297-9716-44-72
- Ryt-Hansen et al., 2022
The role of gilts in transmission dynamics of swine influenza virus and impacts of vaccination strategies and quarantine managementPorc. Health Manag, 8 (2022), p. 19, 10.1186/s40813-022-00261-2
- Salvesen and Whitelaw, 2021
Current and prospective control strategies of influenza A virus in swinePorc. Health Manag, 7 (2021), p. 23, 10.1186/s40813-021-00196-0
- Sandbulte et al., 2015
Optimal use of vaccines for control of influenza A virus in swineVaccin. (Basel), 3 (2015), pp. 22-73, 10.3390/vaccines3010022
- Schaefer et al., 2015
A human-like H1N2 influenza virus detected during an outbreak of acute respiratory disease in swine in BrazilArch. Virol., 160 (2015), pp. 29-38, 10.1007/s00705-014-2223-z
- Schultz-Cherry et al., 2013
History of Swine InfluenzaJ.A. Richt, R.J. Webby (Eds.), Swine Influenza, Springer Berlin Heidelberg, Berlin, Heidelberg (2013), pp. 21-27, 10.1007/82_2011_197
- Snell, 2010
Cost-Benefit Analysis Second edition: A Practical Guide (2nd ed.), Thomas Telford, London, England (2010), 10.1680/cba.41349
- Song et al., 2010
Serological surveillance of influenza A virus infection in swine populations in Fujian province, China: no evidence of naturally occurring H5N1 infection in pigsZoonoses Public Health, 57 (2010), pp. 291-298, 10.1111/j.1863-2378.2009.01270.x
- Toft et al., 2005
A framework for decision support related to infectious diseases in slaughter pig fattening unitsAgric. Syst., 85 (2005), pp. 120-137, 10.1016/j.agsy.2004.07.017
- Torremorell et al., 2012
Transmission of influenza A virus in pigsTransbound. Emerg. Dis., 59 (1) (2012), pp. 68-84, 10.1111/j.1865-1682.2011.01300.x
- United States Department of Agriculture-National Agricultural Statistics Service, 2024
United States Department of Agriculture-National Agricultural Statistics Service, 2024. USDA (2024) 2022 Census of Agriculture, Summary and State Data.
- USDA-APHIS, 2021
USDA-APHIS, 2021. VS Memorandum (VSM) 800.69 [WWW Document]. URL 〈https://www.aphis.usda.gov/sites/default/files/memo800-69.pdf〉 (accessed 1.26.26).
- USDA-APHIS, 2024
USDA-APHIS, 2024. Biosecurity on U.S. Swine Operations NAHMS Swine 2021 Study [WWW Document]. URL 〈https://www.aphis.usda.gov/sites/default/files/swine-large-ent-biosecurity-2021-infobrief.pdf〉 (accessed 1.27.26).
- Vaccination administration via gel saves time, labor and money [WWW Document], 2025
Vaccination administration via gel saves time, labor and money [WWW Document], 2025. URL 〈https://bi-animalhealth.com/swine/articles/vaccination-administration-via-gel-saves-time-labor-and-money〉 (accessed 12.23.25).
- VanderWaal and Deen, 2018
Global trends in infectious diseases of swineProc. Natl. Acad. Sci. U. S. A., 115 (2018), pp. 11495-11500, 10.1073/pnas.1806068115
- Vincent et al., 2008
Failure of protection and enhanced pneumonia with a US H1N2 swine influenza virus in pigs vaccinated with an inactivated classical swine H1N1 vaccineVet. Microbiol, 126 (2008), pp. 310-323, 10.1016/j.vetmic.2007.07.011
- Vincent et al., 2010
Efficacy of inactivated swine influenza virus vaccines against the 2009 A/H1N1 influenza virus in pigsVaccine, 28 (2010), pp. 2782-2787, 10.1016/j.vaccine.2010.01.049
- What and when: Deeper look at wean-to-finish mortality [WWW Document], 2024
What and when: Deeper look at wean-to-finish mortality [WWW Document], 2024. National Hog Farmer. URL 〈https://www.nationalhogfarmer.com/livestock-management/what-and-when-deeper-look-at-wean-to-finish-mortality〉 (accessed 1.27.26).
- White et al., 2017
Influenza A virus in swine breeding herds: Combination of vaccination and biosecurity practices can reduce likelihood of endemic piglet reservoirPrev. Vet. Med., 138 (2017), pp. 55-69, 10.1016/j.prevetmed.2016.12.013
- Wickham et al., 2007
Ggplot2: Create elegant data visualisations using the grammar of graphicsCRAN Contrib. Packages (2007), 10.32614/cran.package.ggplot2
- Wolf and Wolf, 2005
Prod. Livest. Dis. Manag. Incent. Decis. (2005), 10.22004/AG.ECON.8179
- Zhang et al., 2014
An integrated epidemiological and economic analysis of vaccination against highly pathogenic porcine reproductive and respiratory syndrome (PRRS) in thua Thien hue province, VietnamAsianAustralas. J. Anim. Sci., 27 (2014), pp. 1499-1512, 10.5713/ajas.2014.14060
- Zhou et al., 2021
Porcine reproductive and respiratory syndrome modified live virus vaccine: A “leaky” vaccine with debatable efficacy and safetyVaccin. (Basel), 9 (2021), p. 362, 10.3390/vaccines9040362





